Vyaire Recalls AirLife Humidification Chamber & Heated Breathing Circuit Kits
January 30, 2018: Vyaire Medical is a Chicago-based medical device company that focuses on manufacturing respiratory products.
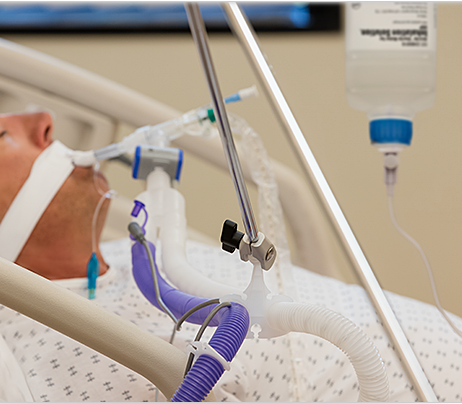
The FDA has announced a Class I recall of Vyaire’s “AirLife Humification Chamber and Heated Breathing Circuit Kits”. A Class I recall is the most serious recall issued by the FDA. It is issued in situations where a product or device may cause serious injury or death.
Vyaire’s AirLife Kits are used in hospitals and other healthcare settings to provide humified breathing gasses for adults and children.
According to the FDA, the medical device is being recalled due to a manufacturing defect that may cause the chamber to split apart, allowing excessive amounts of water to enter the airway or lungs of a ventilated patient. This could lead to serious adverse health consequences, including injury or death.
About 16,670 units are being recalled nationwide. The recalled devices were manufactured and/or distributed between May 18, 2017, and November 10, 2017. Healthcare facilities have been sent an “Urgent Recall Notification” letter instructing them to inspect their inventory and remove and destroy all affected products.
If you or a loved one were on a ventilator and suffered an injury, you should report the matter to MedWatch, the FDA’s online adverse event reporting system.
If you would like to speak to one of our highly skilled medical device team members, please call (877) 230-6690. An attorney and a medical professional evaluate each case and let individuals know what they believe the best course of action to be. All case evaluations and consultations are free and confidential.

